Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 Sr
Sr CoO
CoO at initial electrochemical process in an alkaline solution
at initial electrochemical process in an alkaline solutionMatsuzaki, Akira*; Hirayama, Masaaki*; Oguchi, Shoya*; Komo, Mamoru*; Ikezawa, Atsunori*; Suzuki, Kota*; Tamura, Kazuhisa; Arai, Hajime*; Kanno, Ryoji*
Electrochemistry (Internet), 90(10), p.107001_1 - 107001_8, 2022/10
Times Cited Count:0 Percentile:0.01(Electrochemistry)Oxygen reduction and evolution reactions (ORR and OER) of perovskite-type La Sr
Sr CoO
CoO were characterized using two-dimensional model electrodes with different reaction planes. Synthesized by pulsed laser deposition, these thin and flat electrodes can reveal the reaction plane dependence of the ORR activity. From steady-state polarization measurements in KOH (aq.), the ORR activity was the highest on the (001) film during the first ORR/OER cycle, and it decreased significantly during the second cycle. In-situ synchrotron X-ray diffraction clarified crystal structure changes in the bulk and surface regions of La
were characterized using two-dimensional model electrodes with different reaction planes. Synthesized by pulsed laser deposition, these thin and flat electrodes can reveal the reaction plane dependence of the ORR activity. From steady-state polarization measurements in KOH (aq.), the ORR activity was the highest on the (001) film during the first ORR/OER cycle, and it decreased significantly during the second cycle. In-situ synchrotron X-ray diffraction clarified crystal structure changes in the bulk and surface regions of La Sr
Sr CoO
CoO , and these changes are associated with forming oxygen defects during the initial electrochemical process. Furthermore, the La
, and these changes are associated with forming oxygen defects during the initial electrochemical process. Furthermore, the La Sr
Sr CoO
CoO surface partially decomposed upon reacting. Therefore, the interfacial structures formed in the electrochemical reaction field is important for enhancing ORR and OER activities.
surface partially decomposed upon reacting. Therefore, the interfacial structures formed in the electrochemical reaction field is important for enhancing ORR and OER activities.
 Fe
Fe O
O
Yagi, Yutaro*; Wakita, Yudai*; Kagomiya, Isao*; Matsue, Ikuya*; Kakimoto, Kenichi*; Matsumura, Daiju; Yoneda, Yasuhiro
ChemistrySelect (Internet), 7(21), p.e202104575_1 - e202104575_7, 2022/06
Times Cited Count:0 Percentile:0(Chemistry, Multidisciplinary)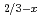 Li
Li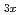 TiO
TiO (x = 0.13) studied by quasielastic neutron scattering
(x = 0.13) studied by quasielastic neutron scatteringMatsuura, Masato*; Fujiwara, Yasuyuki*; Moriwake, Hiroki*; Ohara, Koji*; Kawakita, Yukinobu
Physical Review B, 104(9), p.094305_1 - 094305_7, 2021/09
Times Cited Count:5 Percentile:44.86(Materials Science, Multidisciplinary)Kong, L.*; Gong, J.*; Hu, Q.*; Capitani, F.*; Celeste, A.*; Hattori, Takanori; Sano, Asami; Li, N.*; Yang, W.*; Liu, G.*; et al.
Advanced Functional Materials, 31(9), p.2009131_1 - 2009131_12, 2021/02
Times Cited Count:22 Percentile:80.38(Chemistry, Multidisciplinary)The soft nature of organic-inorganic halide perovskites renders their lattice particularly tunable to external stimuli such as pressure, undoubtedly offering an effective way to modify their structure for extraordinary optoelectronic properties. However, these soft materials meanwhile feature a general characteristic that even a very mild pressure will lead to detrimental lattice distortion and weaken the critical light-matter interaction, thereby triggering the performance degradation. Here, using the methylammonium lead iodide as a representative exploratory platform, we observed the pressure-driven lattice disorder can be significantly suppressed via hydrogen isotope effect, which is crucial for better optical and mechanical properties previously unattainable.
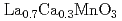
Sato, Wataru*; Komatsuda, Sayaka*; Osa, Akihiko; Sato, Tetsuya; Okubo, Yoshitaka*
Hyperfine Interactions, 237(1), p.113_1 - 113_6, 2016/12
Times Cited Count:2 Percentile:61.78(Physics, Atomic, Molecular & Chemical)The magnetic hyperfine field and electric field gradient at the Cd(
Cd(
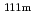 Cd) and
Cd) and 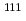 Cd(
Cd(
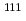 In) probe nuclei introduced in a perovskite manganese oxide
In) probe nuclei introduced in a perovskite manganese oxide 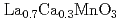 (
(
 250 K) were measured for the study of the local magnetism and structure by means of time-differential perturbed angular correlation spectroscopy. In the ferromagnetic phase at 77 K, a very slight supertransferred magnetic hyperfine field (SMHF) (
250 K) were measured for the study of the local magnetism and structure by means of time-differential perturbed angular correlation spectroscopy. In the ferromagnetic phase at 77 K, a very slight supertransferred magnetic hyperfine field (SMHF) ( 0.014 T) combined with a well-defined electric field gradient was observed at the nonmagnetic
0.014 T) combined with a well-defined electric field gradient was observed at the nonmagnetic 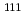 Cd nucleus on the La/Ca A site. This observation suggests that the large magnetic hyperfine field (
Cd nucleus on the La/Ca A site. This observation suggests that the large magnetic hyperfine field (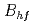 = 6.9 T) measured, in our previous work, at the
= 6.9 T) measured, in our previous work, at the 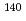 Ce probe nucleus on the A site originates from the contribution of a 4
Ce probe nucleus on the A site originates from the contribution of a 4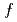 spin oriented by the SMHF from adjacent Mn ions.
spin oriented by the SMHF from adjacent Mn ions.
Miyazawa, Yu*; Ikegami, Masashi*; Miyasaka, Tsutomu*; Oshima, Takeshi; Imaizumi, Mitsuru*; Hirose, Kazuyuki*
Proceedings of 42nd IEEE Photovoltaic Specialists Conference (PVSC-42) (CD-ROM), p.1178 - 1181, 2015/06
 Ca
Ca MnO
MnO (
(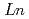 =Ho, Er, Tm, Yb and Lu)
=Ho, Er, Tm, Yb and Lu)Yoshii, Kenji; Abe, Hideki*; Ikeda, Naoshi*
Journal of Solid State Chemistry, 178(12), p.3615 - 3623, 2005/12
Times Cited Count:25 Percentile:67.33(Chemistry, Inorganic & Nuclear)no abstracts in English
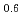 Ru
Ru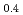 O
O
Yoshii, Kenji; Nakamura, Akio; Mizumaki, Masaichiro*; Tanida, Hajime*; Kawamura, Naomi*; Abe, Hideki*; Ishii, Yoshinobu; Shimojo, Yutaka; Morii, Yukio
Journal of Magnetism and Magnetic Materials, 272-276(Suppl.), p.e609 - e611, 2004/05
Times Cited Count:4 Percentile:25.12(Materials Science, Multidisciplinary)no abstracts in English
Kawamura, Yoshinori; Konishi, Satoshi; Nishi, Masataka
Fusion Science and Technology, 45(1), p.33 - 40, 2004/01
Times Cited Count:25 Percentile:81.94(Nuclear Science & Technology)Aiming at realization of the efficient blanket tritium recovery system of a fusion reactor, research and development of the hydrogen pump using the proton conductor are furthered. One of the advantages of the system using the hydrogen pump is concurrent processing of hydrogen isotopes and water vapor by one component. In this work, experimental research on the hydrogen extraction characteristic in a water molecule was performed with the hydrogen pump using SrCe Yb
Yb O
O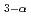 . In hydrogen extraction from a water molecule, application of the threshold, voltage corresponding to the decomposition energy of water, is necessary. The observed threshold was about 500-600mV at 873K and decreased with the increase in water vapor pressure. About pumping of H
. In hydrogen extraction from a water molecule, application of the threshold, voltage corresponding to the decomposition energy of water, is necessary. The observed threshold was about 500-600mV at 873K and decreased with the increase in water vapor pressure. About pumping of H -H
-H O mixture gas, the permeation of H
O mixture gas, the permeation of H anteceded water decomposition, and the threshold of water decomposition increased with the increase in hydrogen partial pressure. In order to process concurrently, application of fairly higher voltage is expected to be necessary.
anteceded water decomposition, and the threshold of water decomposition increased with the increase in hydrogen partial pressure. In order to process concurrently, application of fairly higher voltage is expected to be necessary.
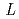
 Sr
Sr CoO
CoO (
(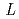 =La, Pr, Nd, Sm, and Eu)
=La, Pr, Nd, Sm, and Eu)Yoshii, Kenji; Abe, Hideki*
Physical Review B, 67(9), p.094408_1 - 094408_8, 2003/03
Times Cited Count:40 Percentile:82.47(Materials Science, Multidisciplinary)no abstracts in English
 Dy
Dy TiO
TiO
Yoshii, Kenji; Mizumaki, Masaichiro*; Nakamura, Akio; Abe, Hideki*
Journal of Solid State Chemistry, 171(1-2), p.345 - 348, 2003/02
Times Cited Count:9 Percentile:30.79(Chemistry, Inorganic & Nuclear)no abstracts in English
Nishihata, Yasuo; Tanaka, Hirohisa*
SPring-8 Riyosha Joho, 7(6), p.359 - 363, 2002/11
Catalytic converters are widely applied to control automotive emissions, such as nitrogen oxides, carbon monoxide and unburned hydrocarbons. In actual catalysts, however, the catalytic activity deteriorates due to the particle growth of precious metals during vehicle use. An ageless catalyst is a kind of philosopher's stone in automotive engineering. Referring to the wisdom of ancient Indian philosophy and medical science, Ayur Veda, we have developed a new self-regenerating perovskite-based catalyst. The perovskite catalyst, LaFe Co
Co Pd
Pd O
O , regenerates itself without any auxiliary treatment, in direct reaction to the natural fluctuation between reductive and oxidative atmospheres in the exhaust gas from state-of-the-art gasoline engines. As palladium reversibly moves back and forth between inside and outside of the perovskite crystal, the particle growth of palladium can be suppressed. Such a self-regeneration provides new insight into the development of future automotive catalyst, as an intelligent catalyst.
, regenerates itself without any auxiliary treatment, in direct reaction to the natural fluctuation between reductive and oxidative atmospheres in the exhaust gas from state-of-the-art gasoline engines. As palladium reversibly moves back and forth between inside and outside of the perovskite crystal, the particle growth of palladium can be suppressed. Such a self-regeneration provides new insight into the development of future automotive catalyst, as an intelligent catalyst.
 V
V O
O
Yoshii, Kenji; Abe, Hideki*
Journal of Alloys and Compounds, 343(1-2), p.199 - 203, 2002/09
Times Cited Count:7 Percentile:47.03(Chemistry, Physical)no abstracts in English
 Sr
Sr CoO
CoO
Mizumaki, Masaichiro*; Yoshii, Kenji; Kawamura, Naomi*; Nakazawa, Makoto*
Surface Review and Letters, 9(2), p.855 - 859, 2002/04
Times Cited Count:1 Percentile:7.78(Chemistry, Physical)no abstracts in English

Yoshii, Kenji; Abe, Hideki*
Physica B; Condensed Matter, 312-313(1-4), p.791 - 792, 2002/03
Times Cited Count:6 Percentile:34.98(Physics, Condensed Matter)no abstracts in English
 Sr
Sr CoO
CoO (Ln=Pr, Nd, Sm, Eu and Gd)
(Ln=Pr, Nd, Sm, Eu and Gd)Yoshii, Kenji; Nakamura, Akio; Abe, Hideki*; Mizumaki, Masaichiro*; Muro, Takayuki*
Journal of Magnetism and Magnetic Materials, 239(1-3), p.85 - 87, 2002/02
Times Cited Count:15 Percentile:59.47(Materials Science, Multidisciplinary)no abstracts in English
Yoshii, Kenji; Nakamura, Akio
Recent Research Developments in Physics, 3, p.758 - 776, 2002/00
no abstracts in English
 Pr
Pr CrO
CrO
Yoshii, Kenji; Nakamura, Akio; Ishii, Yoshinobu; Morii, Yukio
Journal of Solid State Chemistry, 162(1), p.84 - 89, 2001/11
Times Cited Count:100 Percentile:96.55(Chemistry, Inorganic & Nuclear)no abstracts in English

Yoshii, Kenji
Journal of Solid State Chemistry, 159(1), p.204 - 208, 2001/08
Times Cited Count:189 Percentile:99.17(Chemistry, Inorganic & Nuclear)no abstracts in English
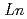
 Sr
Sr CoO
CoO (
(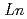 =Pr, Nd, Sm and Eu)
=Pr, Nd, Sm and Eu)Yoshii, Kenji; Abe, Hideki*; Nakamura, Akio
Materials Research Bulletin, 36(7-8), p.1447 - 1454, 2001/06
Times Cited Count:41 Percentile:79.02(Materials Science, Multidisciplinary)no abstracts in English